Research in the Van Vranken group is focused on the prediction of products and mechanisms using deep learning and the development of powerful reactions for the synthesis of bioactive compounds, mostly involving either transition metal catalysts or peptide substrates.
1. Powerful Tools for Arrow-Pushing Mechanisms
Our group specializes in chemoinformatic tools for prediction of organic reaction products and the arrow-pushing mechanisms by which organic products form. We recently completed the first quantitation of the nucleophilicities and electrophilicities of the common organic functional groups based on methyl ion affinity. Armed with an understanding of fundamental reactivity and an expansive knowledge of arrow-pushing mechanisms, we are working with experts in a specialized form of machine learning to predict mechanistic steps and multi-step mechanistic pathways.
2. New Synthetic Methods
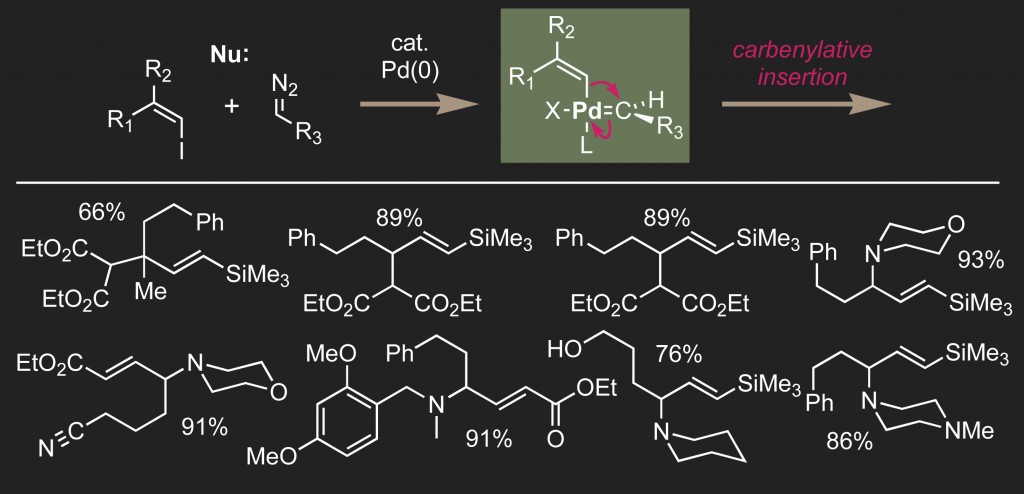
Most of our synthetic efforts are now centered on metal-catalyzed carbenylative insertion processes. We have developed palladium-catalyzed carbenylative cross-coupling reactions that generate vinylsilanes, gamma-amino acids, and allylamines, providing one-step access to key structures that are common in a variety of natural products.
We have demonstrated the ability to vary the carbene donor, the halide acceptor and nucleophile in palladium-catalyzed carbenylative cross-couplings. With the ability to vary each individual component of the reaction we are well-positioned to synthesize a wide range of complex molecules, including natural products and bioactive compounds.
Potential Targets
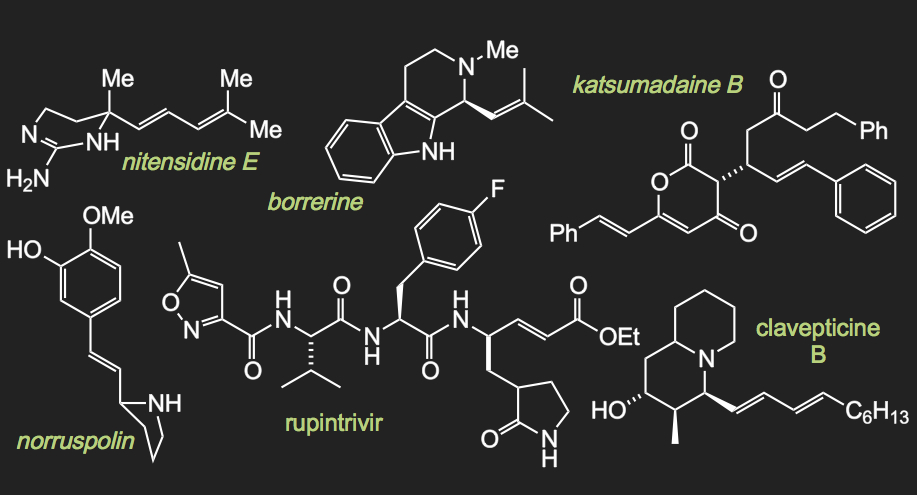
Our recent success in the development of metal-catalyzed carbenylative insertion reactions should allow us to extend the list of natural products successfully synthesized by our group.
Natural Products Synthesized by the Van Vranken Group